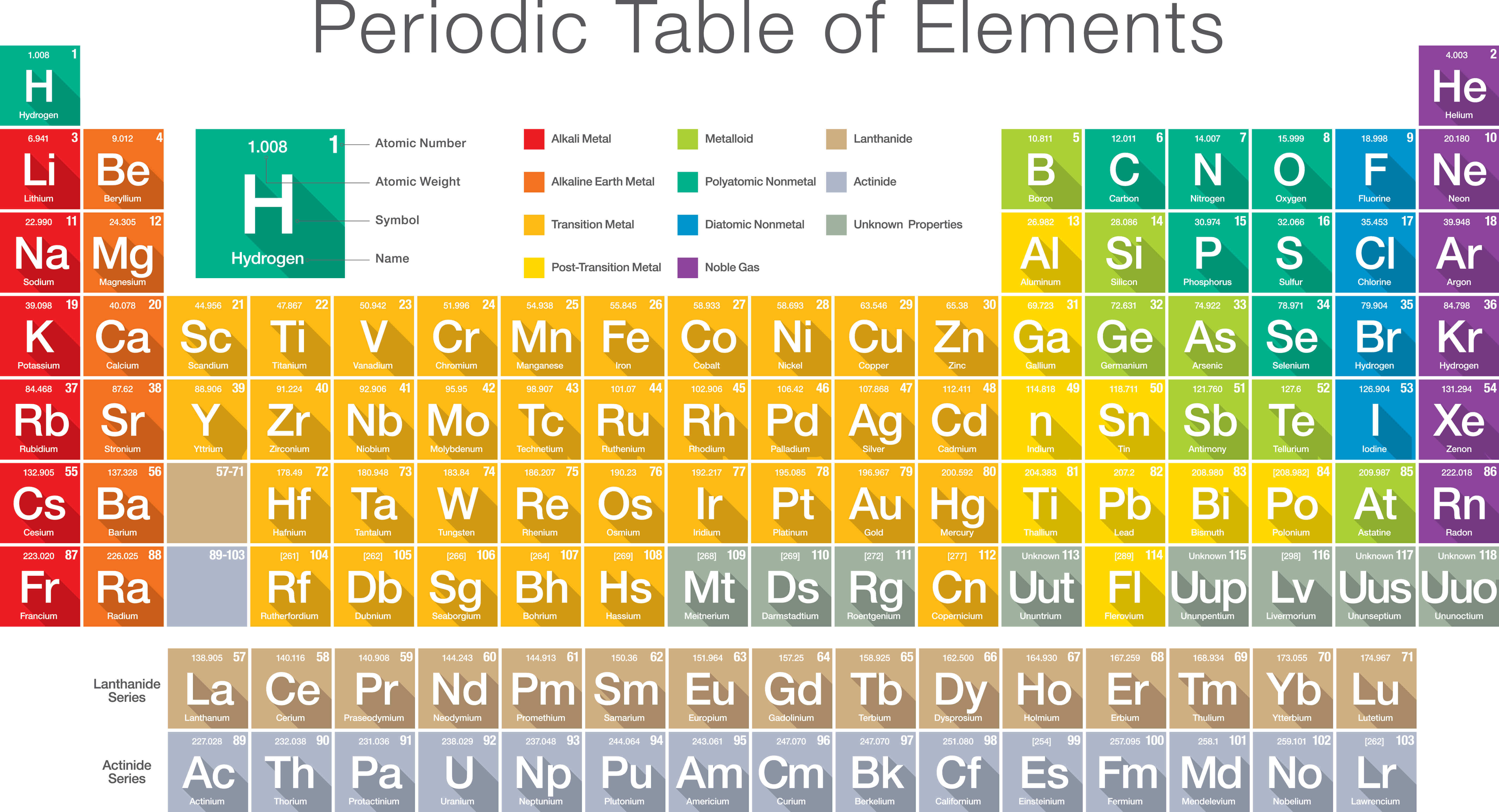 
IUPAC, European, and American Group labelling schemes Group The last set (main group elements A, transition elements B) was in common use in America. The first of these (A left, B right) is based upon older IUPAC recommendations and frequently used in Europe. The designations A and B are completely arbitrary. The other two systems are less desirable since they are confusing, but still in common usage. The scheme used in WebElements is numeric and is the current IUPAC convention. There is considerable confusion surrounding the Group labels. Image showing periodicity of group numbers for group 13 chemical elements. In addition, groups may be idenitifed by the first element in each group - so the Group 16 set of elements is sometimes called the oxygen group. Group 15: pnictogens (not an IUPAC approved name).Group 11: coinage metals (not an IUPAC approved name).The following names for specific groups in the periodic table are in common use: The second row of the f -block elements are called actanoids (or, less desirably, actanides. 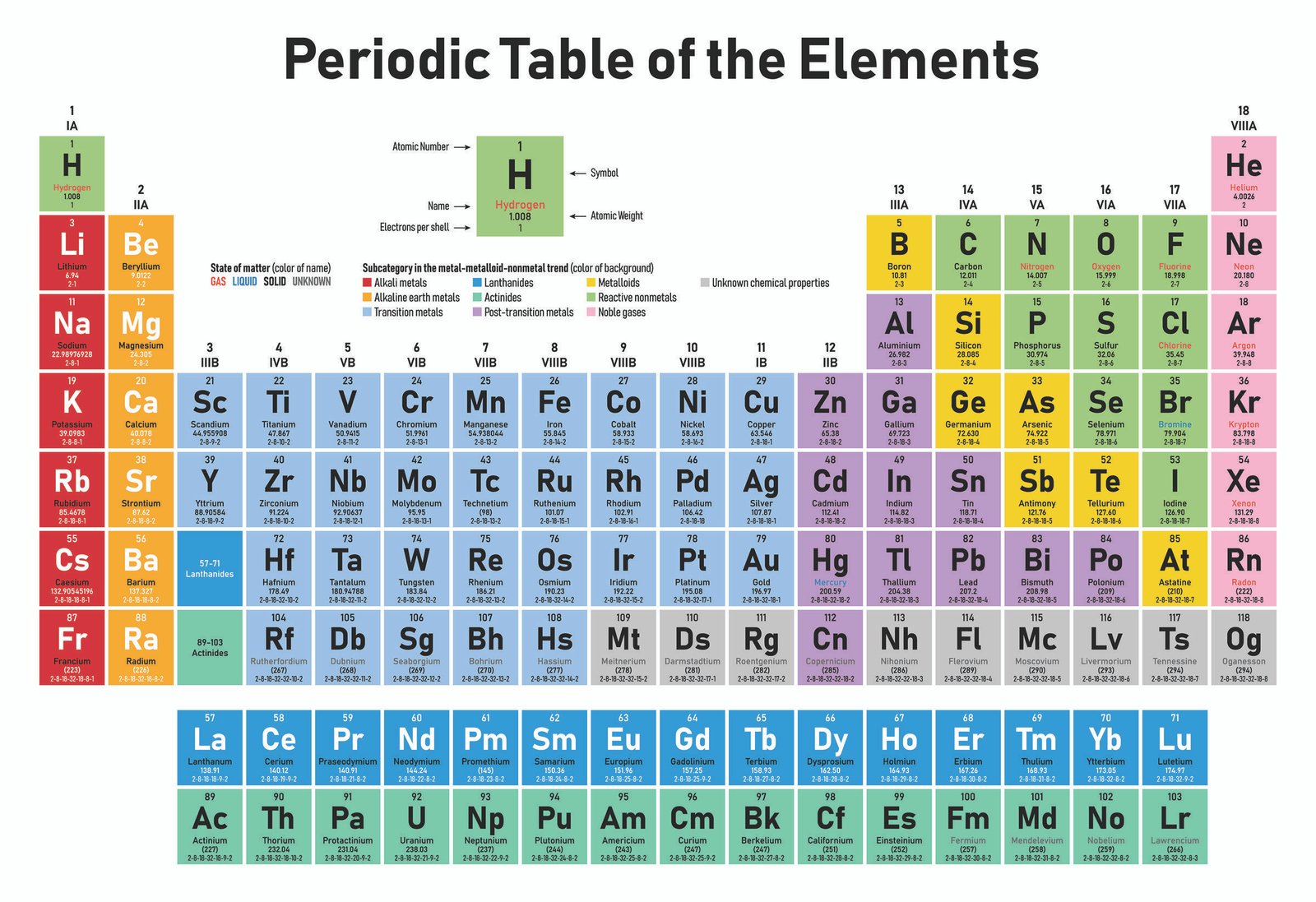
The first row of the f-block elements are called lanthanoids (or, less desirably, lanthanides. Main group elements in the first two rows of the table are called typical elements. Transition elements are those whose atoms have an incomplete d-subshell or whose cations have an incomplete d-subshell. Groups 3-11 are termed transition elements. Groups 1-2 (except hydrogen) and 13-18 are termed main group elements. The group number is an identifier used to describe the column of the standard periodic table in which the element appears.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
